AP Chemistry: Electron Configuration
AP Chemistry: Electron configuration. This article explains how electrons are arranged outside the nucleus, from shells to subshell notation, and why electron configuration matters for understanding atomic structure and chemical behavior.
Authors: SHSBNU Chem Club
AP Chemistry: Electron Configuration
AP Chemistry
Electrons Outside the Nucleus
Electron configuration is a compact way to describe how electrons are arranged around an atom. Once we understand shells, subshells, orbitals, and the main filling rules, many later topics in chemistry become much easier.
Quick Review: Electron Shells
Electrons around the nucleus are arranged in layers according to energy. The closest layer is the first shell, which can hold at most 2 electrons. Farther shells can hold more.
The number of electrons in the outermost shell strongly affects chemical behavior:
- metals usually lose electrons more easily
- nonmetals often gain electrons more easily
- noble gases are especially stable when their outer shell is filled
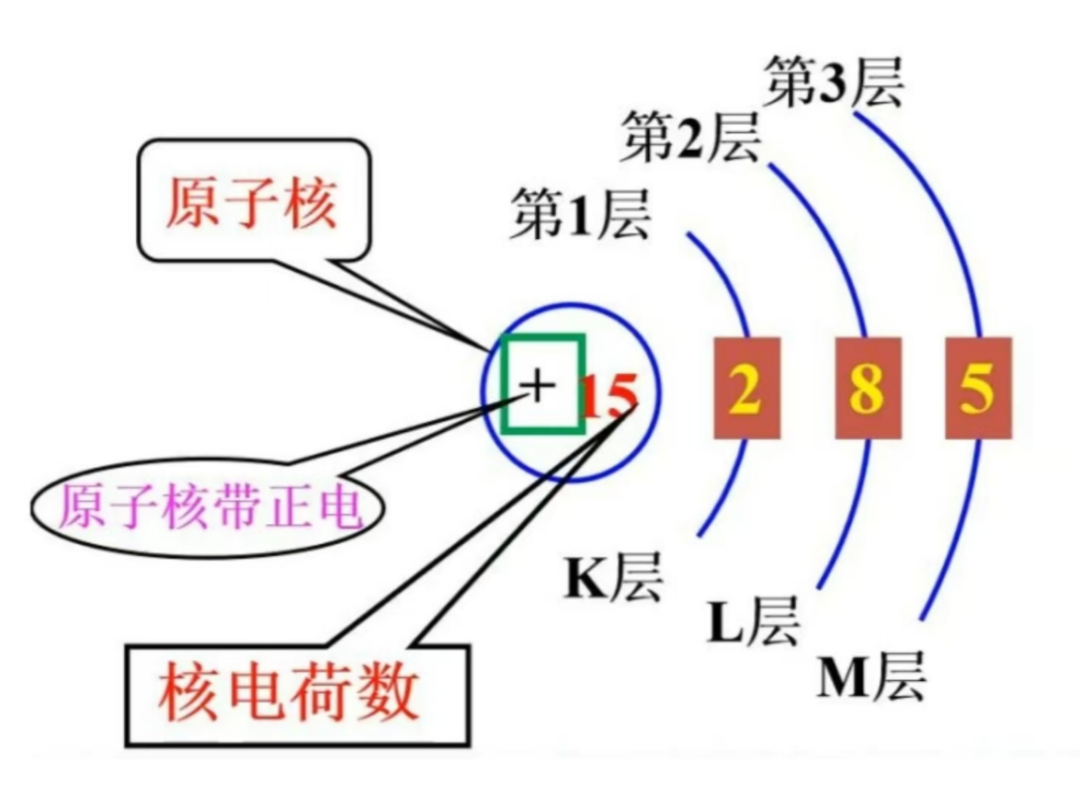
An atom in its lowest-energy arrangement is in the ground state. If one of its electrons is promoted to a higher energy level, the atom enters an excited state.
Shells and Subshells
In AP Chemistry, we go beyond the simple shell picture.
The shell number is written as n. If n = 3, for example, the electron belongs to the third principal energy level. Higher shells generally correspond to higher electron energy.
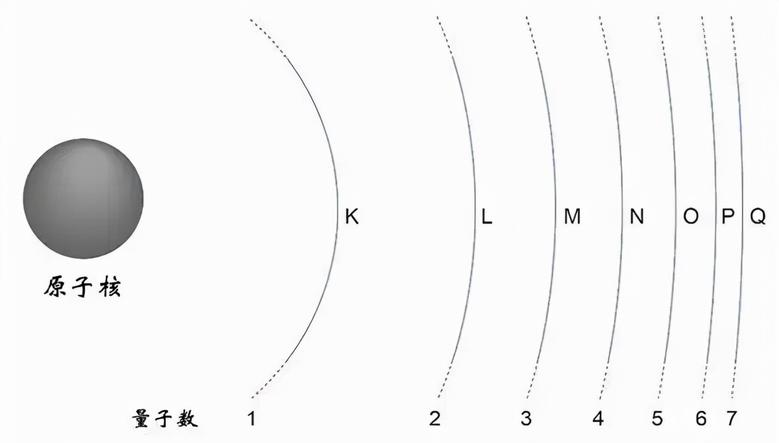
The maximum number of electrons in shell n is 2n^2.
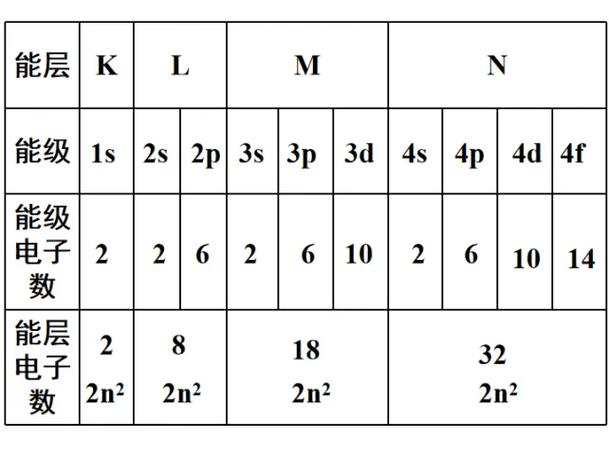
But electrons are not described only by shell. Each shell is divided into subshells, or energy sublevels, identified by letters:
spdf
Within the same shell, these subshells do not all have the same energy:
s < p < d < f

Orbitals and Capacities
Each subshell contains a fixed number of orbitals:
s: 1 orbitalp: 3 orbitalsd: 5 orbitalsf: 7 orbitals
Every orbital can hold at most 2 electrons, so the maximum capacities are:
s: 2 electronsp: 6 electronsd: 10 electronsf: 14 electrons
Some arrangements are especially stable. Fully filled, half-filled, and completely empty subshells can be favored, especially in p, d, and f sets. That is why chromium and copper do not follow the simplest expected filling pattern:
- Cr is better written as
4s1 3d5 - Cu is better written as
4s1 3d10
Similar patterns also appear in elements such as Mo and Ag.
Spin, Hund's Rule, and the Pauli Principle
Two electrons in the same orbital must have opposite spins.
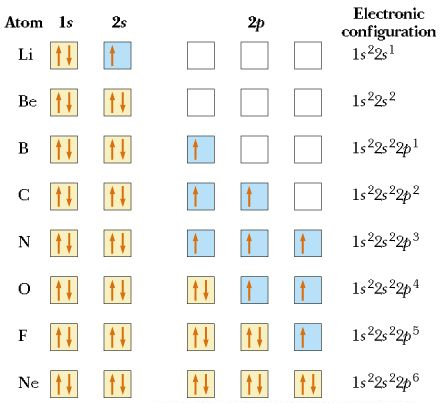
When orbitals have the same energy, electrons occupy them singly with parallel spins before pairing up. This is Hund's rule.
At this stage we can summarize four important quantum numbers:
nlmlms
No two electrons in the same atom can have exactly the same complete set of quantum numbers. This is the Pauli exclusion principle.
Writing Electron Configurations
Because of energy-level overlap, some orbitals in a higher shell can actually be lower in energy than orbitals in a lower shell. That is why we do not fill orbitals simply by shell number.
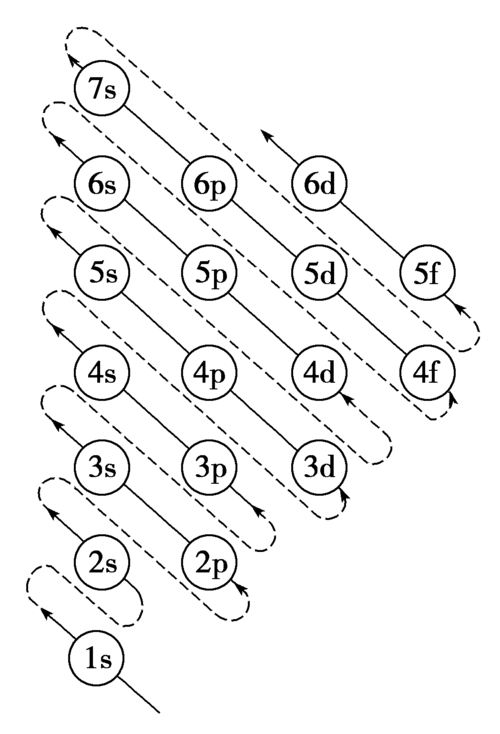
When writing electron configurations, we follow the order of increasing orbital energy.
Valence electrons are the electrons that can interact with other atoms to form chemical bonds.
As a practical guide:
- for
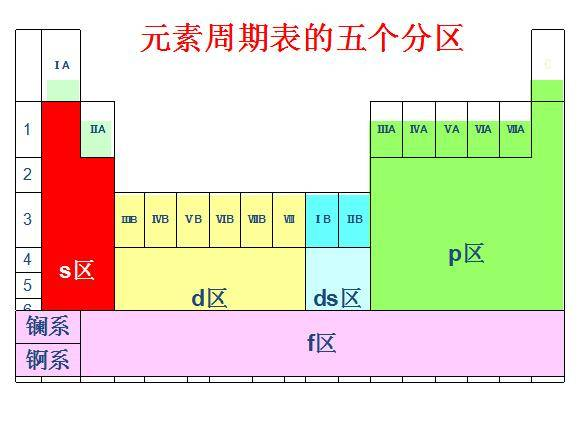
s- andp-block elements, valence electrons are the electrons in the outermost shell - for
d-block elements, valence electrons include the outer-shell electrons and electrons in a higher-energy inner subshell - AP Chemistry usually does not require a full treatment of
f-block valence electrons
This article is best used as a preview or review sheet. Once the ideas are clear, the next step is to practice writing configurations and solving problems regularly.
