The Final Experiment of 2025: Elephant Toothpaste
Our final experiment of the year: Elephant Toothpaste.
Authors: SHSBNU Chem Club
The Last Experiment of 2025
Our final experiment of the year__: Elephant Toothpaste
Principle
2H2O2 -> 2H2O + O2
Potassium iodide can be used as a catalyst in this reaction. A catalyst lowers the activation energy, so hydrogen peroxide decomposes much faster.
If KI is used, the reaction pathway can be illustrated like this:
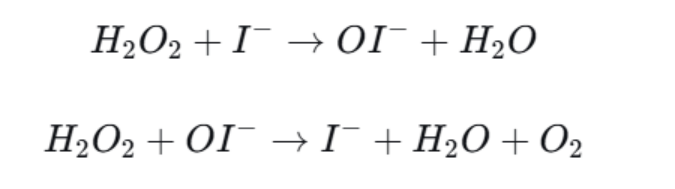
The catalyst keeps the same chemical identity before and after the reaction, so it can be reused.
Why Add Dish Soap?
If hydrogen peroxide decomposed on its own, we would mostly just see oxygen bubbles rise and disappear. The spectacular "toothpaste" effect comes from dish soap.
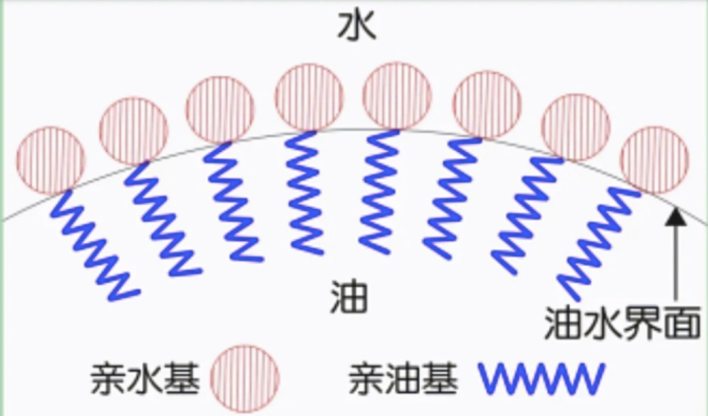
Dish soap contains surfactants that trap gas and stabilize foam. A surfactant has a hydrophilic end that prefers water and a hydrophobic end that avoids it. When oxygen is produced, the hydrophobic ends gather around the bubbles while the hydrophilic ends stay in the surrounding water. That creates a molecular film on the bubble surface, and countless bubbles stack together into the dense foam we see.
Experiment Photos



Reagents and Equipment
Reagents:
- 20 mL dish soap
- glucose
- 10 mL glycerol
- 60 mL of 30% hydrogen peroxide
- potassium iodide or manganese dioxide
- food coloring
Equipment:
- beaker
- Erlenmeyer flask
- magnetic stirrer
Procedure
- Prepare solution A: mix 20 mL dish soap with a small amount of glucose and 10 mL glycerol, stir well, then add 60 mL of 30% H2O2. Food coloring can be added if desired.
- Prepare solution B: dissolve 2-3 g potassium iodide in 10 mL warm water.
- Add solution A to the Erlenmeyer flask first, then pour in solution B.
Here is the experiment video:
END
