Copper Sulfate Roses: Crystal Blossoms from Solution
When crystals grow on roses, ordinary petals turn into dazzling blue blooms. This article introduces the copper sulfate rose experiment and the chemistry of crystal growth.
Authors: SHSBNU Chem Club
Copper Sulfate Rose
When Crystals Bloom on Fabric
2025 JUN.12TH
SHSBNU Chem Club

Experiment Overview
When crystals grow on a cloth flower, the petals begin to sparkle. In this activity, we prepared a supersaturated copper sulfate solution and introduced a seed for crystallization. The excess solute quickly began to come out of solution and grow on the rose. By gently changing the flower's position, the crystals could grow more evenly. After drying, the flower became a blue "crystal rose."



Why It Works
Supersaturated Solution
A supersaturated solution is an unstable state in which the solute concentration exceeds its normal solubility at a given temperature and pressure. It can exist temporarily, but even a small disturbance such as stirring, vibration, or the introduction of a seed crystal can break the balance and trigger rapid crystallization.
Crystals and Crystal Cells
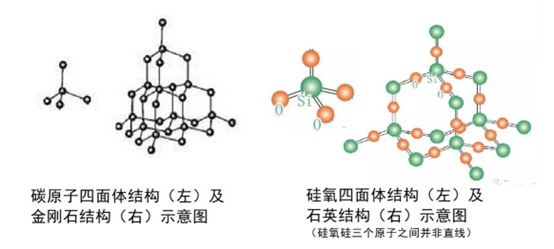
A crystal is a solid formed by atoms, molecules, or ions arranged periodically in three-dimensional space. The smallest repeating unit is called a unit cell, and its geometry determines much of the crystal's symmetry and macroscopic shape.
Examples of familiar crystals include ice, quartz, and diamond.


Chemists also classify crystals according to the dominant type of chemical bonding:
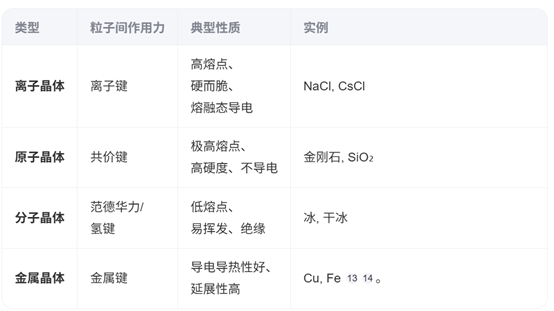
Crystal materials appear throughout modern life, from integrated circuits and semiconductors to optical devices.
Procedure
Step 1: Prepare a Supersaturated Copper Sulfate Solution
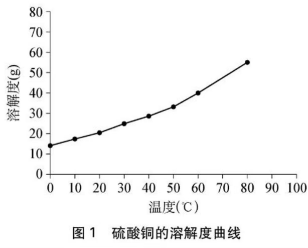
Add 140 g copper sulfate to a beaker, then add distilled water to a total volume of 180 mL. Heat the mixture to about 90 C to prepare a saturated solution. Continue heating until the solution becomes supersaturated, then stop heating and let it cool to about 80 C.


Step 2: Put the Rose into the Solution
Place the fabric rose into the solution and wait for nucleation to begin. Let initial crystals attach to the back of the flower, then use a straw to slowly guide the flower deeper into solution and encourage crystal growth on the petals. After enough growth, remove the flower, turn it upside down, and let it dry.

Highlights


