What Is an Atom? A Journey into the Microscopic World
From Dalton's solid sphere to Thomson's plum-pudding model, Bohr's quantized orbits, and Schrödinger's electron cloud, this article traces how our picture of the atom has been continually rewritten.
Authors: SHSBNU Chem Club
What Is an Atom?
A Guided Tour Through the Microscopic World
From Dalton's solid sphere, to Thomson's plum-pudding model, to Bohr's quantized orbits and Schrodinger's electron cloud, our picture of the atom has been rewritten again and again.
Each new model marked a deeper breakthrough in humanity's attempt to understand matter. This article follows some of those major turning points.
Part 1: John Dalton
John Dalton (1766-1844) was a British chemist and physicist. He is best known for proposing atomic theory, which transformed chemistry from the study of visible change into a science with microscopic explanation.

Dalton noticed that substances react in fixed proportions. For example, hydrogen and oxygen combine in a constant ratio when forming water. He concluded that matter must be made of tiny particles, each element having its own kind of atom and its own characteristic mass.
From these observations he proposed that:
- all matter is made of atoms
- atoms of the same element share the same properties and mass
- atoms are tiny, indivisible solid particles
- compounds form when atoms combine in fixed ratios

Part 2: J. J. Thomson
J. J. Thomson (1856-1940) was born near Manchester in England. Through experiments with cathode rays, he measured the charge-to-mass ratio of the particles in the beam and showed that atoms contain negatively charged components: electrons.

This led him to propose the plum-pudding model. In that picture, the atom is a positively charged sphere with electrons embedded inside it, like raisins in pudding. The model was not the final answer, but it was the first major step beyond Dalton's indivisible atom.
Part 3: Niels Bohr
Niels Bohr (1885-1962), the Danish theoretical physicist, proposed a new atomic model in 1913 based on Rutherford's nuclear atom.
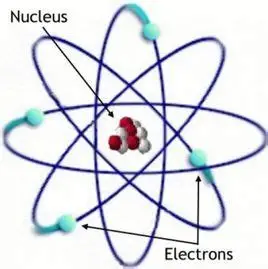

Bohr introduced three important ideas:
- Electrons can move in certain stable orbits around the nucleus without radiating energy.
- Electron angular momentum is quantized.
- Electrons can jump between allowed orbits only by absorbing or emitting electromagnetic radiation of specific energies.
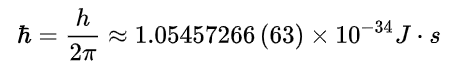
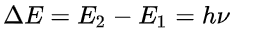
Bohr's model successfully explained the stability of the atom and the spectral lines of hydrogen. It also pushed quantum theory forward dramatically.
Part 4: Erwin Schrodinger
Erwin Schrodinger (1887-1961) was an Austrian physicist and Nobel Prize laureate. He helped build modern quantum mechanics.

Schrodinger's equation became one of the foundational laws of quantum mechanics, playing a role similar to Newton's laws in classical mechanics. He also proposed the famous Schrodinger's cat thought experiment to challenge how quantum ideas are interpreted at the macroscopic scale.
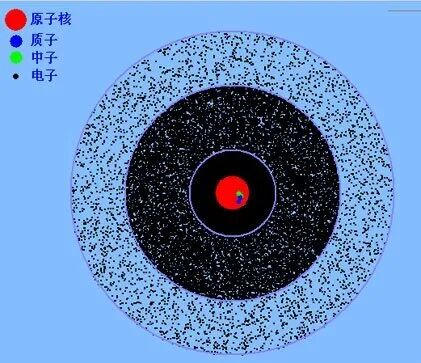
In the quantum picture, electrons are no longer tiny planets circling the nucleus along simple tracks. Instead, they are described by probability distributions, often visualized as electron clouds. Together with the uncertainty principle, this changed our picture of the atom from a mechanical orbit system into a probabilistic quantum object.
