Neanderthals and Chemistry
Neanderthals lived in Europe and western Asia tens of thousands of years ago. This article explores the chemistry behind their tools, fire use, and painting materials.
Authors: SHSBNU Chem Club
Neanderthals and Chemistry
Neanderthals (Homo neanderthalensis) lived in Europe and western Asia roughly 120,000 to 30,000 years ago. Fossils show that they adapted to environments ranging from temperate regions to extremely cold climates.
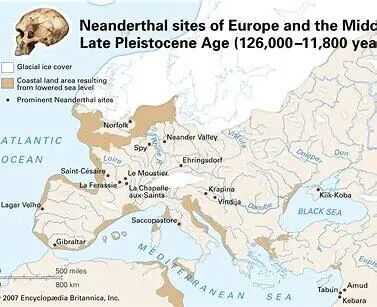
They relied heavily on large mammals such as mammoths, rhinoceroses, and deer, and isotope analysis suggests that animal protein made up more than 80% of their diet. They also gathered plant foods such as berries, nuts, and tubers, especially when game was scarce.


Neanderthals disappeared around 40,000 years ago under a combination of pressures: small effective population size, inbreeding risk, climate instability, competition with Homo sapiens, and gradual genetic dilution through limited interbreeding. Even so, during their long history they demonstrated surprisingly sophisticated practical chemistry.
Toolmaking

Archaeologists found traces of birch-bark tar on stone tools from Neanderthal sites near the modern border of Italy and Germany. As early as 200,000 years ago, Neanderthals were using adhesive substances to fix stone blades onto wooden handles.
When birch bark is heated, it releases a sticky tar-like resin that can bind wood, leather, and stone together. That means Neanderthals were not only using materials - they were transforming them through controlled heating.
Firemaking


Neanderthals also used manganese dioxide to help start fires. At a site in southwestern France, scientists found stones with abrasion marks. When the material was ground into powder and sprinkled onto wood, the ignition temperature dropped from about 350 C to about 250 C.
Why Does Manganese Dioxide Help?
The key idea is catalysis.

The term "catalyst" was introduced by the Swedish chemist Jons Jacob Berzelius. A catalyst changes the rate of a chemical reaction without being permanently consumed and without changing the overall equilibrium of the reaction.
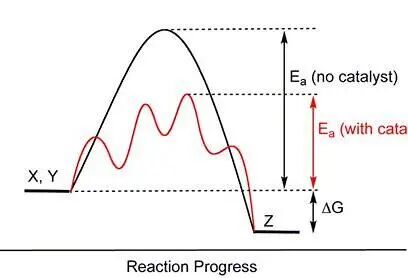
Catalysts lower activation energy, so less heat is needed to make the reaction proceed. That is why wood could ignite more easily in the presence of manganese dioxide.
Activation energy is the energy required to move reactants from their normal state into a more reactive state.
At high temperatures, manganese dioxide can also release oxygen during decomposition. Because oxygen is essential for combustion, that extra oxygen can help the flame burn more intensely.
