"From Stone to Blue": The Chemistry of Ultramarine
This article explores the chemistry behind ultramarine, the historic blue pigment seen in art, and explains its composition, color, and cultural significance.
Authors: SHSBNU Chem Club
Chemistry in Art
Ultramarine: Blue Born from Stone
Perhaps you have seen Girl with a Pearl Earring. The luminous blue headscarf in that painting contrasts sharply with the dark background and helps create its quiet, melancholy mood.

Where does that bright, pure blue come from? The answer is ultramarine.
Ultramarine was originally produced by grinding lapis lazuli, a precious blue stone, into powder. It is also an inorganic blue pigment made from sulfur, clay, quartz, carbon, and related raw materials through high-temperature firing.


Its formula is written here as Na6Al4Si6S4O. Ultramarine belongs to a crystal family based on a sodalite-type framework. In simple terms, the structure can be understood as two parts:
- a rigid framework built from silicon, aluminum, and oxygen
- active ions such as sodium and sulfur trapped in cage-like cavities
Structure
The rigid framework is made of Si4+ and Al3+ linked through O2- atoms. These tetrahedral units connect into a stable three-dimensional network.
The cavity sites contain sodium ions and sulfur-based species. Sodium balances charge, while sulfur species play a major role in color and some chemical behavior.
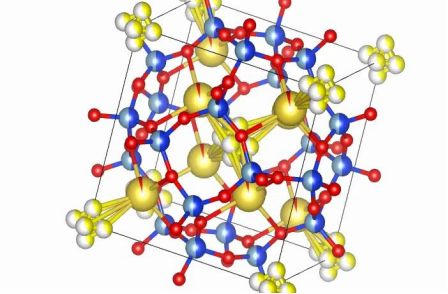
This structure helps explain why ultramarine is durable. The cage-like framework protects sulfur species from air, oxygen, and ultraviolet light, which is why the pigment can keep its color for long periods. It is also insoluble in water and many organic solvents, helping it remain stable in paints and plastics.
Its resistance is not unlimited, though. Strong oxidizing acids such as concentrated nitric acid can break the structure down and release sulfur-containing products.
Why Is Ultramarine Blue?
The blue color is also controlled by structure. Sulfur-containing species inside the cages undergo electronic transitions whose energies are fixed by the local environment of the crystal.
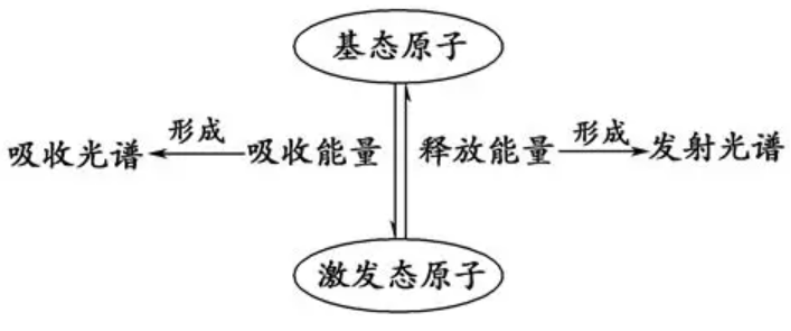
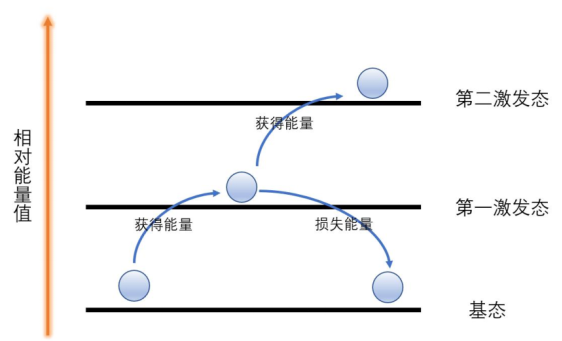
Electrons cannot absorb just any energy. They can only absorb photons whose energy exactly matches the gap between allowed levels. In ultramarine, sulfur-centered species absorb orange-yellow light particularly well, so the reflected light that reaches our eyes is blue.
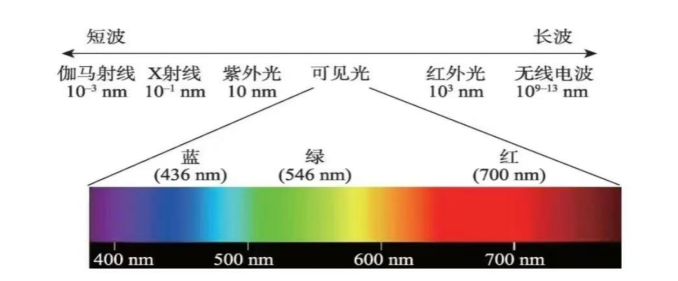
Why Was It So Expensive?

Ultramarine was once called "royal blue" or even "blue gold." In medieval Europe it became a symbol of status and prestige.
One reason was scarcity: natural ultramarine came from rare lapis lazuli deposits, and mining and processing were costly. Even synthetic ultramarine still requires energy-intensive firing of raw materials such as quartz, soda ash, and sulfur, so manufacturing carries a real cost barrier.
