Iron's Dual Identity
Is iron more commonly +2 or +3, and why? Starting from electron configuration and Hund’s rule, this article explains the two common oxidation states of iron in chemistry and daily life.
Authors: SHSBNU Chem Club

Iron's Dual Identity
Why Can Iron Be +2 or +3?
Many students notice that some elements, such as Fe, Cu, or Mn, often appear in more than one oxidation state. Why is that? Iron is a great example.
Basic Facts About Iron

- atomic number: 26
- electron configuration:
1s2 2s2 2p6 3s2 3p6 3d6 4s2or[Ar] 3d6 4s2 - relative atomic mass: 55.847
Iron's First Identity: +2
The outermost part of a neutral iron atom contains two 4s electrons. Those two electrons are lost most easily, producing Fe2+ with the configuration [Ar] 3d6.
Common examples include FeO and FeCl2.


Iron's Second Identity: +3


Starting from Fe2+, the 3d subshell still contains six electrons. That is not one of the especially stable patterns. In many transition metals, half-filled or fully filled subshells are relatively stable. For a d subshell, a half-filled arrangement is d5.
So after iron loses one more electron, it becomes Fe3+ with a 3d5 arrangement, which is often more stable than 3d6.
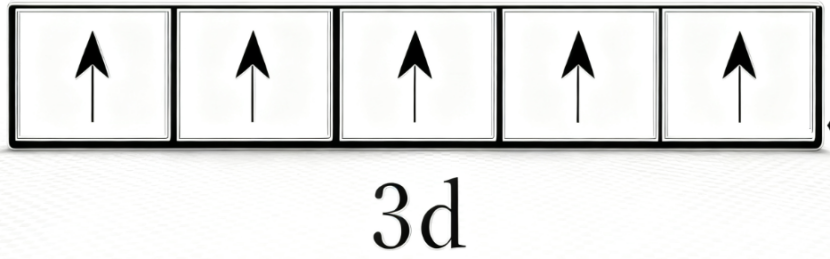
Hund's Rule
Why is the d5 pattern favorable? This is where Hund's rule helps.

Friedrich Hund proposed that electrons in orbitals of equal energy prefer to occupy them singly, with parallel spins, before pairing up. That lowers the total energy of the system.
Fe3+ matches this especially well in the five d orbitals, while Fe2+ already has one paired electron in the d set and is therefore somewhat easier to oxidize to Fe3+ under suitable conditions.
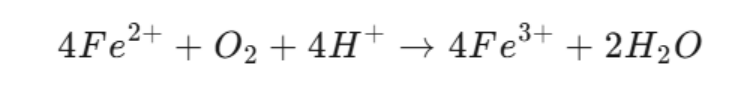
Examples of Fe3+ compounds include Fe2O3 and FeCl3.


Iron in Daily Life
Iron's two common oxidation states appear everywhere around us:
- Fe2+ occurs in iron supplements such as ferrous sulfate and is important for plant chlorophyll synthesis.
- Fe3+ is a major component of rust and contributes to the reddish-brown color of many soils.
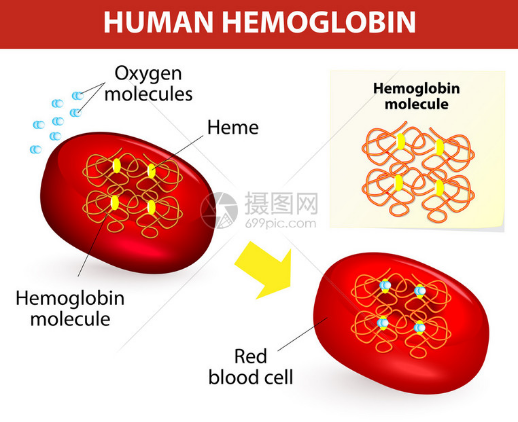
- The Fe2+ / Fe3+ redox pair also participates in electron transfer in living systems, including hemoglobin- and cytochrome-related processes.