"A Garden in Water": Plant-Like Growth in Alkaline Solution
Water gardens are classic chemistry demonstrations in which metal-salt crystals grow into colorful, plant-like branching structures in solution.
Authors: SHSBNU Chem Club

Water Garden
"Plants" Growing in Alkaline Solution
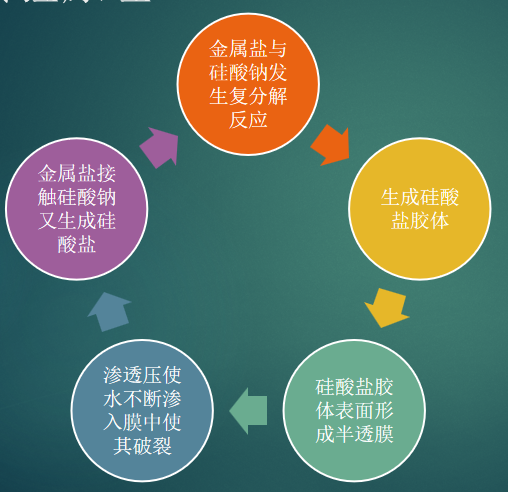
The water garden is a classic chemistry demonstration. When metal-salt crystals are dropped into a suitable solution, they grow into colorful branching shapes that resemble underwater plants. The key ideas are semipermeable membrane formation, osmotic pressure, chemical reaction, and crystal growth.
Sodium silicate solution provides the alkaline environment for this experiment. Because it is a clear, colorless, viscous liquid and can dry into a glass-like solid, it is also known as "water glass."
In this activity, the club used copper sulfate, nickel chloride, zinc chloride, magnesium chloride, ferric nitrate, and calcium chloride crystals as the "plants." These crystals repeatedly form a membrane, swell under osmotic pressure, rupture, and regrow, producing more and more branches until an underwater garden appears.
Steps
- Spread a layer of clean fine sand about 1 cm thick on the bottom of a beaker.
- Dissolve 10 g sodium silicate in 40 mL water.
- Use tweezers to place 3-5 mm crystals of copper sulfate, nickel chloride, zinc chloride, magnesium chloride, ferric nitrate, calcium chloride, and similar salts at different spots on the sand in the sodium silicate solution.
- Wait and observe. Blue, green, white, yellow, and other bud-like or tree-like structures gradually grow from the crystals.

Notes
- Do not shake or disturb the beaker, because vibration can break the silicate gel structures.
- Try to distribute the crystals evenly when placing them into the solution.
Here is a video from the club experiment:
