"Heart of the Ocean": Changes in Chromium Oxidation States
"Heart of the Ocean" is a striking color-change experiment. By adding hydrogen peroxide and dilute sulfuric acid to potassium chromate solution, the colors rapidly shift through yellow, orange, blue, and green, revealing chromium in different oxidation states.
Authors: SHSBNU Chem Club
Heart of the Ocean
Color Changes of Chromium in Different Oxidation States

"Heart of the Ocean" is a striking color-change demonstration. By adding hydrogen peroxide and dilute sulfuric acid to potassium chromate solution, the liquid rapidly changes from yellow to orange, then blue, and finally green. The experiment vividly shows the characteristic colors of chromium in different oxidation states.
Chromium Colors
Hexavalent chromium appears in two common forms:
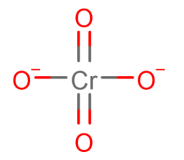
- chromate is yellow
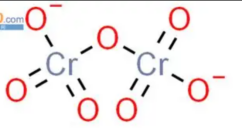
- dichromate is orange
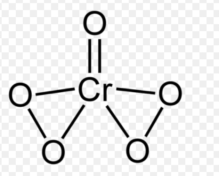
Peroxochromium, often written as CrO5 in this context, appears blue:
Trivalent chromium ions are green.
Reaction Principle
- Chromate reacts with hydrogen ions to form dichromate and water.
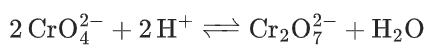
- Dichromate reacts with hydrogen peroxide to form peroxochromium (CrO5) and water.
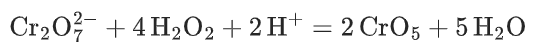
- Under acidic conditions, peroxochromium is reduced more quickly to Cr3+. The oxygen generated in the process is also why bubbles appear during the experiment.
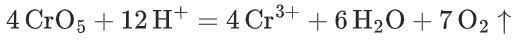
Materials
- beakers
- graduated cylinder
- spatula
- dropper
- potassium chromate
- dilute sulfuric acid
- hydrogen peroxide solution
Procedure
- Add 0.1 g potassium chromate to beaker A, then add 150 mL water.
- Add about one third of a dropper of hydrogen peroxide solution.
- Add 15 mL dilute sulfuric acid to beaker B.
- Transfer a small amount of solution from beaker A into beaker B and observe the color change.
Process Photos


Final Result



