Flame Tests
Why do different metals produce different colors in a flame? This article explains flame tests from the perspective of spectra and energy transitions.
Authors: SHSBNU Chem Club
Flame Tests
Have you ever wondered why different metals seem to "speak" in different colors when heated in a flame? Flame tests are a great way to connect visible color with spectra, electronic structure, and energy transitions.
Principle
The essence of a flame test is energy absorption and release by electrons in metal ions.
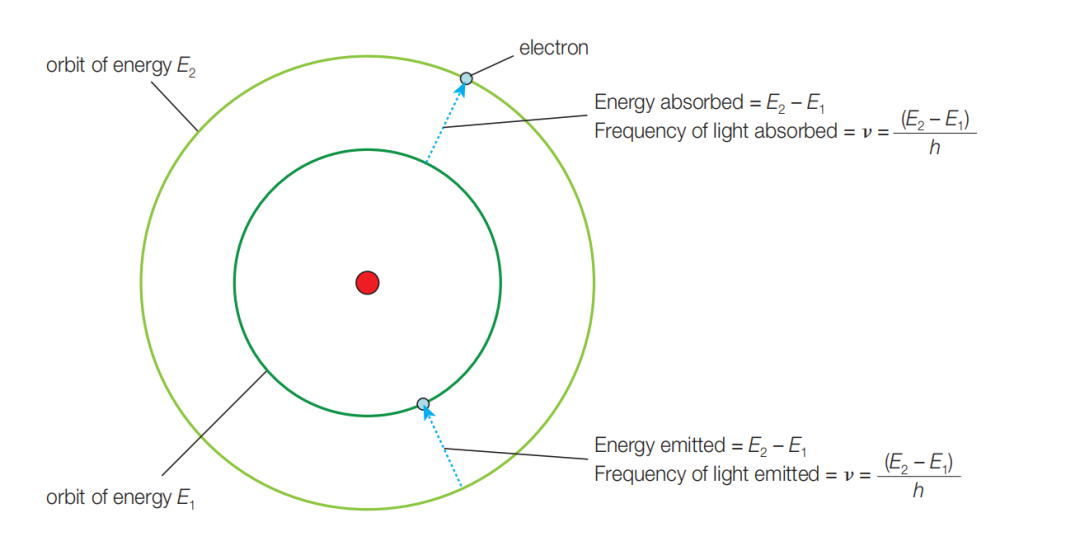
When a metal ion is heated in a flame, electrons in the outer region absorb energy and jump from the ground state to an excited state. This stores energy temporarily.
Excited electrons are unstable, so they quickly fall back to lower energy levels. The excess energy is released as photons, and that light is what we see as flame color.
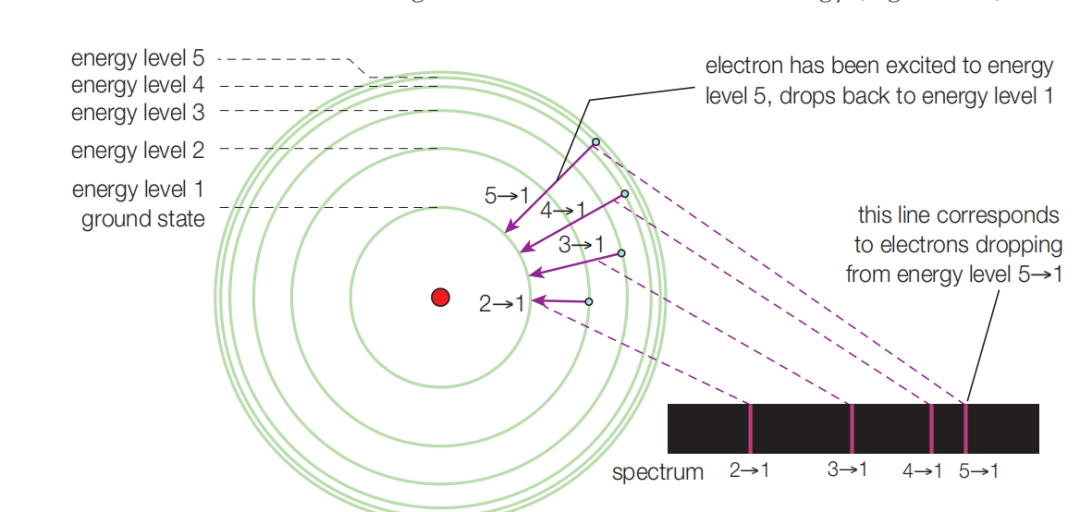
Because different metals have different electron arrangements and different energy gaps, they emit light of different wavelengths. Our eyes interpret those wavelengths as different colors.
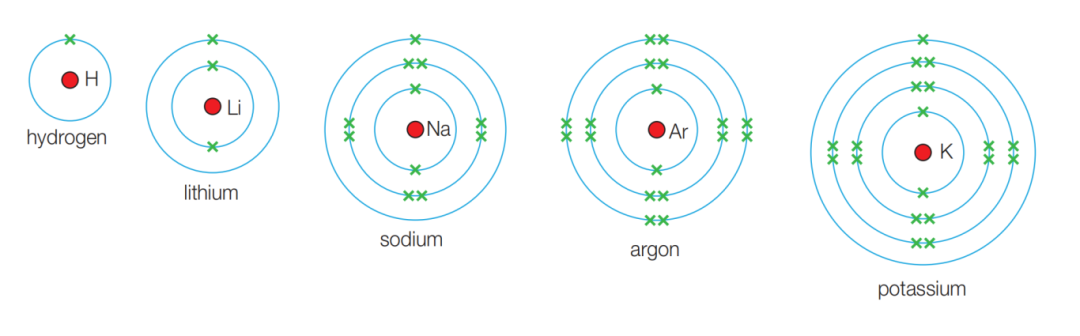
Common examples:
- sodium: bright yellow, around 589 nm
- potassium: pale lilac, often easier to observe through cobalt glass
- copper: blue-green, around 510 nm
- lithium: crimson
- calcium: brick red

Procedure
- Add 20 mL distilled water to a beaker.
- Dissolve an appropriate amount of metal salt to make a saturated solution. Boric acid forms a supersaturated suspension instead.
- Use a dropper to transfer about 2-3 mL of the saturated salt solution and mix it with an equal volume of ethanol in an evaporating dish.
- Hold the incense stick at the end, keep your arm extended, and use it to ignite the liquid in the evaporating dish.
- Remove the incense stick immediately after ignition and secure it safely.
- Make sure the evaporating dish cools completely before beginning the next trial.
Solubility of Selected Metal Salts at 20 C
- sodium chloride: 35.9 g / 100 mL
- potassium chloride: 34.2 g / 100 mL
- calcium chloride: 70.0-74.5 g / 100 mL
- strontium chloride hexahydrate: 52.9 g / 100 mL
- lithium chloride: 83.5 g / 100 mL
- boric acid: 5 g / 100 mL
- barium chloride: 35.8 g / 100 mL
- copper chloride: 77.3 g / 100 mL
