Green Chemistry: Acid Rain Series, Part I
Part I of our green chemistry series on acid rain.
Authors: SHSBNU Chem Club
Green Chemistry
Acid Rain Series, Part I
SHSBNU Chem Club
After the unification of Germany in 1871, heavy industry played a major role in rapid economic growth. But the environmental cost was severe. Large areas of forest were damaged, and by the early 1980s many trees in West Germany had already suffered widespread decline. In places such as Bavaria, Hesse, and Hamburg, forest damage became a major warning sign. One important cause behind this destruction was acid rain.

Distribution of Acid Rain in China
China became one of the world's major acid-rain regions after Europe and North America. In the 1980s, the main affected areas were represented by Chongqing, Guiyang, and Liuzhou. By the mid-1990s, the affected area had expanded widely across regions south of the Yangtze River, east of the Qinghai-Tibet Plateau, and across the Sichuan Basin.
Central China became one of the most seriously affected areas, with some core regions experiencing acid rain in more than 90% of rainfall events. Eastern coastal cities such as Nanjing, Shanghai, Hangzhou, Fuzhou, Qingdao, and Xiamen also became important acid-rain zones. In some places, acid rain covered more than 30% of China's land area.
Why Acid Rain Forms
Acid rain is usually defined as precipitation with pH < 5.6. It is a by-product of highly industrialized society. When large amounts of coal, oil, and natural gas are burned, sulfur oxides and nitrogen oxides are released into the air. These gases undergo complex atmospheric chemistry and eventually form sulfuric acid or nitric acid aerosols, or become dissolved in cloud droplets, rain, snow, or fog.
Pathway 1: Sulfur Oxides to Sulfuric Acid
Sulfur dioxide (SO2) is produced mainly by coal-fired power plants and industrial boilers.
Once in the atmosphere, SO2 can dissolve in cloud droplets or raindrops and form sulfurous acid. Oxidants such as ozone and hydrogen peroxide then convert it into sulfuric acid (H2SO4).
Step 1: Dissolution
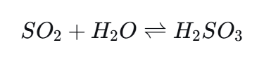
Step 2: Oxidation to Sulfuric Acid
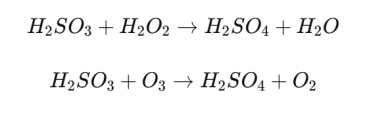
Step 3: Ionization
Sulfuric acid is a strong acid. In water it ionizes extensively and releases hydrogen ions.
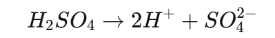
Pathway 2: Nitrogen Oxides to Nitric Acid
Nitrogen oxides (NOx) mainly include nitric oxide (NO) and nitrogen dioxide (NO2). These gases come largely from vehicle exhaust and high-temperature combustion.
Gas-Phase Chemistry
High-temperature combustion produces large amounts of NO, which is then oxidized further in air.
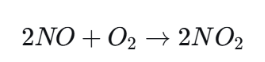
NO2 can react with the highly reactive hydroxyl radical (.OH) in the atmosphere to form nitric acid.

Liquid-Phase Chemistry
Nitrogen dioxide can also dissolve in tiny droplets in air or clouds and form nitric acid there.
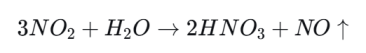
Nitric acid is also a strong acid. Once dissolved in water, it releases hydrogen ions and makes rainfall strongly acidic.
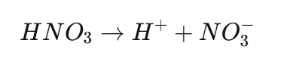
END
Contributors
唐泽霖
Role: Contributor
Focus: Writing, Review
辛敏睿
Role: Contributor
Focus: Writing
尉钰涵
Role: Contributor
Focus: Writing, Layout
孟禹萌
Role: Contributor
Focus: Writing
张若凡
Role: Contributor
Focus: Review
